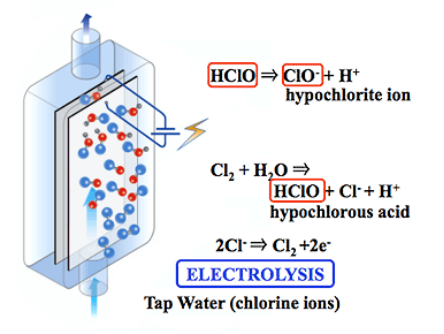
Chlorine is the sum of the three forms of Cl2, HClO and ClO-, and the ratio of HCl and ClO- is determined by the pH value of the solution. When the solution solution contains Cl-, hypochlorous acid or hypochlorite is produced, and Cl- is oxidized to produce Cl2 (Cl2), Cl2 raw water forms hypochlorous acid and hydrochloride (2), hypochlorous acid and hypochlorite In the dynamic equilibrium between chlorates (Equations 4, 3), the relative amount of heat varies with the pH of the human body. The whole process is regenerated. Therefore, when the solution is circulated, the chemical composition of the solution is basically unchanged.
2Cl-→Cl2+2e-(1)
Cl2 + H2O → HClO + HCl (2)
HClO〈==〉ClO- + H+ (3)
HClO → O + Cl- + H+ (4)
ClO- → O + Cl- (5)
