
Experimental report of sodium hypochlorite
Xi’an Qinhuang Intelligent Co., LTD
Tel:029-89388827
M :17782629907
http://www.qinhuangwater.com
Email:athur@qinhuangwater.com
Add:No.8, Gaoxin 3rd Road, High-Tech Zone, Xi’an, Shaanxi, China 710075
Ⅰ Title: electrolysis of 3%~5% brine to produce sodium hypochlorite.
Ⅱ Experimental date: November 24 to December 31, 2019.
Ⅲ Objective: to change the experimental conditions and get the best effective chlorine solution in the best time
Ⅳ Experimental principle: using titanium plate coated with precious metal coating as anode, direct current electrolysis of low concentration salt water (3% ~ 5%), electrode electrochemical reaction to produce sodium hypochlorite solution.
Reaction formula: NaCl + H2O → NaClO + H2 ↑
Electrode reaction: anode 2Cl–– 2e → Cl2
Cathode 2H + + 2e → H2
Solution reaction: 2NaOH + Cl2 → NaCl + NaClO + H2O
Ⅴ Materials and methods
Materials:
- Electrode: provided by Qinhuang Intelligent Technology Co., Ltd. (100 mm * 50 mm * 1 mm, 30 mm * 30 mm * 1 mm, 35 mm * 35 mm * 1 mm, 40 mm * 40 mm * 1 mm), MAGNETO special anodes . (100 mm * 50 mm * 1 mm, 30 mm * 30 mm * 1 mm, 35 mm * 35 mm * 1 mm, 40 mm * 40 mm * 1 mm), ruthenium iridium double-sided coating;
- Power supply: Zhaoxin rxn-3020a DC regulated power supply;
- Detection tablets: palintest dpd1 and detection box (the detection range of available chlorine is 0.1-6.0mg/l);
- Balance: Shanghai Cenda Jinma JPT-1;
- Shu Niu glass instrument (measuring cylinder, glass rod, conical flask, beaker, etc.);
- Table salt: China Salt Yulin Salt Chemical Co., Ltd. (iodine 20 ~ 50mg / kg, selenium 4.5 ~ 10.7mg / kg).

Methods: a certain amount of salt was completely dissolved in tap water, and the electrode pieces were clamped in a shallow groove with insulating material (fixed electrode spacing). Constant voltage or constant current power supply conditions were set for electrolysis. The original temperature, voltage, current and the available chlorine content after every 10 minutes were recorded (the detection range of the detection box was 0.1-6.0 mg / L, while the available chlorine content of the general generated solution was 1000-8000 mg / L, which was more than 1000 times, Therefore, it is necessary to test after dilution. The method is to absorb 0.1 ~ 1ml of the generated solution with a syringe, completely dissolve it in 50 ~ 500ml of water, inject it into the test box, place the tablet, and then compare the color after shaking, and record the effective chlorine content.)
Ⅵ Experimental Result
A Qinhuang30mm*30mm*1mm Anode
1、Under the condition of constant voltage 7V
| electrode space | 8mm | Salt concentration | 30g/l | |||||
| Time | 0 | 10min | 20min | 30min | 40min | 50min | 60min | 70min |
| Voltage V | 7 | 7 | 7 | 7 | 7 | 7 | 7 | 7 |
| Current A | 0.9 | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 | 0.9 | 1.0 |
| Temp.℃ | 20 | 24 | 25 | 26 | 27 | 29 | 29 | 33 |
| Available chlorine | ppm | 400 | 1500 | 1700 | 2000 | 3000 | 3000 | |
2、Under constant current of 1.5A
| electrode space | 8mm | Salt concentration | 30g/l | |||||
| Time | 0 | 10min | 20min | 30min | 40min | 50min | 60min | 70min |
| Voltage V | 8.5 | 8.7 | 8.5 | 8.2 | 8.1 | 8.0 | 8.0 | |
| Current A | 1.5 | 1.5 | 1.5 | 1.5 | 1.5 | 1.5 | 1.5 | |
| Temp.℃ | 17 | 23 | 28 | 32 | 35 | 38 | 40 | |
| Available chlorine | ppm | 400 | 2000 | 4000 | 4000 | 4000 | ||
3、Under constant current of 0.7A
| electrode space | 8mm | Salt concentration | 40g/l | |||||
| Time | 0 | 10min | 20min | 30min | 40min | 50min | 60min | 70min |
| Voltage V | 8 | 7.9 | 8.0 | 8.0 | 7.9 | 7.8 | 7.6 | 7.3 |
| Current A | 0.7 | 0.7 | 0.7 | 0.7 | 0.7 | 0.7 | 0.7 | 0.7 |
| Temp.℃ | 6 | 9 | 12 | 15 | 18 | 19 | 20 | 22 |
| Available chlorine | ppm | 600 | 1000 | 2000 | 2000 | 3000 | 4000 | |
4、Under constant current of 1.0A
| electrode space | 8mm | Salt concentration | 40g/l | |||||
| Time | 0 | 10min | 20min | 30min | 40min | 50min | 60min | 70min |
| Voltage V | 10 | 9.9 | 9.1 | 8.9 | 8.7 | 8.5 | 8.6 | |
| Current A | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | |
| Temp.℃ | 9 | 15 | 20 | 24 | 29 | 33 | 37 | |
| Available chlorine | ppm | 1500 | 3000 | 4000 | 5000 | 5500 | ||
5、Under the condition of constant voltage 8V
| electrode space | 8mm | Salt concentration | 40g/l | |||||
| Time | 0 | 10min | 20min | 30min | 40min | 50min | 60min | 70min |
| Voltage V | 8 | 8 | 8 | 8 | 8 | 8 | 8 | |
| Current A | 0.6 | 0.5 | 0.5 | 0.6 | 0.6 | 0.7 | 0.8 | |
| Temp.℃ | 5 | 10 | 12 | 13 | 15 | 17 | 19 | |
| Available chlorine | ppm | 400 | 1000 | 1500 | 2000 | 3000 | ||
The above experimental curves are as follows:
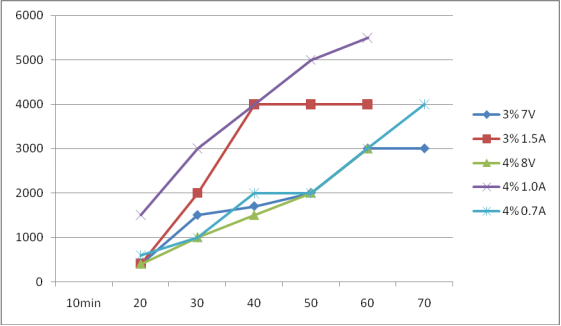
Note: in the 4% 8V experiment, because the weather is cold and the initial water temperature is only 5 ℃, the electrolysis is slow, which affects the effective chlorine value.
B Qinhuang35mm*35mm*1mm Anode
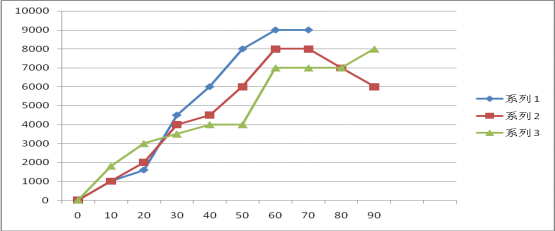
1、Under the condition of constant voltage 8V
| electrode space | 8mm | Salt concentration | 30g/l | ||||||||
| Time | 0 | 10 | 20 | 30 | 40 | 50 | 60 | 70 | 80 | 90 | |
| Voltage V | 8 | 8 | 8 | 8 | 8 | 8 | 8 | 8 | 8 | 8 | |
| Current A | 1 | 1 | 1.2 | 1.2 | 1.2 | 1.4 | 1.4 | 1.9 | 1.9 | 2.0 | |
| Temp.℃ | 8 | 20 | 24 | 28 | 34 | 39 | 43 | 48 | 51 | 56 | |
| Available chlorine | ppm | 1300 | 4000 | 5000 | 5000 | 6000 | 7500 | 6500 | 7500 | ||
2、Under the condition of constant voltage 7V
| electrode space | 8mm | Salt concentration | 40g/l | ||||||||
| Time | 0 | 10 | 20 | 30 | 40 | 50 | 60 | 70 | 80 | 90 | |
| Voltage V | 7 | 7 | 7 | 7 | 7 | 6.9 | 6.8 | 6.8 | 6.8 | 6.8 | |
| Current A | 0.8 | 0.8 | 0.9 | 1.0 | 1.1 | 1.1 | 1.2 | 1.2 | 1.3 | 1.3 | |
| Temp.℃ | 6 | 12 | 15 | 18 | 22 | 25 | 29 | 31 | 35 | 38 | |
| Available chlorine | ppm | 1200 | 3000 | 3000 | 5000 | 6000 | 6000 | 6500 | 7500 | 8000 | |
3、Under the condition of constant voltage 7V
| electrode space | 8mm | Salt concentration | 30g/l | ||||||||
| Time | 0 | 10 | 20 | 30 | 40 | 50 | 60 | 70 | 80 | 90 | |
| Voltage V | 7 | 7 | 6.9 | 6.9 | 6.8 | 6.8 | 6.8 | 6.7 | 6.7 | 6.7 | |
| Current A | 1.1 | 1.1 | 1.2 | 1.2 | 13 | 1.3 | 1.3 | 1.4 | 1.4 | 1.5 | |
| Temp.℃ | 19 | 22 | 25 | 27 | 30 | 33 | 36 | 38 | 40 | 42 | |
| Available chlorine | ppm | 700 | 2000 | 3500 | 3000 | 5000 | 6000 | 6500 | 6500 | 7000
|
|
4、Under the condition of constant voltage 8V
| electrode space | 8mm | Salt concentration | 40g/l | ||||||||
| Time | 0 | 10 | 20 | 30 | 40 | 50 | 60 | 70 | 80 | 90 | |
| Voltage V | 8 | 8 | 8 | 8 | 8 | 8 | 7.9 | 7.9 | 7.9 | 7.9 | |
| Current A | 1.0 | 1.1 | 1.3 | 1.7 | 1.8 | 1.8 | 1.8 | 2.0 | 2.0 | 2.0 | |
| Temp.℃ | 11 | 15 | 23 | 27 | 32 | 39 | 45 | 48 | 48 | 50 | |
| Available chlorine | ppm | 600 | 1600 | 3600 | 3500 | 6000 | 7500 | 6500 | 7000 | 8000 | |
The above experimental curves are as follows:

C Qinhuang40mm*40mm*1mm Anode
1、Under the condition of constant voltage 8V
| electrode space | 6mm | Salt concentration | 40g/l | ||||||
| Time | 0 | 10min | 20min | 30min | 40min | 50min | 60min | 70min | |
| Voltage V | 8 | 8 | 8 | 7.9 | 7.9 | 7.9 | 7.7 | 7.6 | |
| Current A | 1.8 | 1.9 | 2.0 | 2.2 | 2.6 | 2.9 | 3.0 | 3.1 | |
| Temp.℃ | 11 | 16 | 22 | 30 | 37 | 48 | 54 | 59 | |
| Available chlorine | ppm | 1000 | 1600 | 4500 | 6000 | 8000 | 9000 | 9000
|
|
2、Under the condition of constant voltage 8V
| electrode space | 6mm | Salt concentration | 30g/l | |||||||
| Time | 0 | 10 | 20 | 30 | 40 | 50 | 60 | 70 | 80 | 90 |
| Voltage V | 8 | 8 | 8 | 8 | 8 | 8 | 7.9 | 7.9 | 7.9 | 7.9 |
| Current A | 1.7 | 1.7 | 2 | 2.1 | 2.2 | 2.2 | 2.5 | 2.8 | 2.9 | 2.9 |
| Temp.℃ | 11 | 16 | 22 | 29 | 35 | 41 | 46 | 51 | 55 | 58 |
| Available chlorine | ppm | 1000 | 2000 | 4000 | 4500 | 6000 | 8000 | 8000 | 7000 | 6000 |
3、Under the condition of constant voltage 7V
| electrode space | 6mm | Salt concentration | 30g/l | |||||||
| Time | 0 | 10 | 20 | 30 | 40 | 50 | 60 | 70 | 80 | 90 |
| Voltage V | 7.0 | 7.0 | 7.0 | 7.0 | 6.9 | 6.9 | 6.9 | 6.8 | 6.8 | 6.8 |
| Current A | 1.5 | 1.5 | 1.5 | 1.7 | 1.8 | 1.8 | 1.8 | 2.0 | 2.0 | 2.0 |
| Temp.℃ | 11 | 21 | 24 | 27 | 29 | 32 | 35 | 38 | 40 | 43 |
| Available chlorine | ppm | 1800 | 3000 | 3500 | 4000 | 4000 | 7000 | 7000 | 7000 | 8000 |
The above experimental curves are as follows:
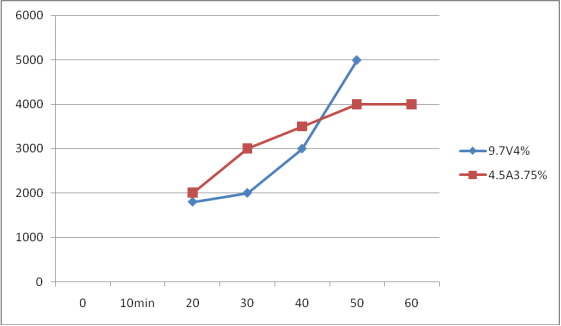
D Qinhuang100mm*50mm*1mm Anode
1、Under the condition of constant voltage 7V
| electrode space | 12mm | Salt concentration | 37.5g/l | |||||
| Time | 0 | 10min | 20min | 30min | 40min | 50min | 60min | 70min |
| Voltage V | 9 | 9 | 9 | 8.5 | 8.4 | 8.3 | ||
| Current A | 4.5 | 5 | 5.1 | 5.3 | 5.7 | 5.8 | ||
| Temp.℃ | 12 | 15 | 23 | 28 | 34 | 38 | ||
| Available chlorine | ppm | 2000 | 3000 | 3500 | 4000 | 4000 | ||
2、Under the condition of constant Current 4.5A
| electrode space | 12mm | Salt concentration | 40g/l | |||||
| Time | 0 | 10min | 20min | 30min | 40min | 50min | 60min | 70min |
| Voltage V | 9.7 | 9.3 | 9 | 8.8 | 8.0 | 7.2 | 7.2 | 7.1 |
| Current A | 4.5 | 4.4 | 4.2 | 4.2 | 4.3 | 4.4 | 4.4 | 4.5 |
| Temp.℃ | 9 | 11.5 | 15 | 22 | 28 | 30 | 35 | 37 |
| Available chlorine | ppm | 1800 | 2000 | 3000 | 5000 | |||
In this experiment, on November 30, the available chlorine was 10000 ppm in 70 minutes, but on December 2, it was only 4000 ppm, so it was not recorded.
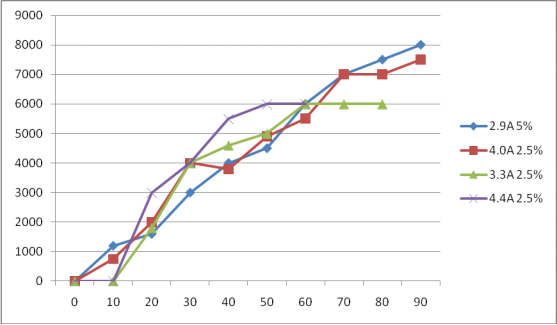
E MAGNETO 100mm*50mm*1mm Anode
According to the experimental conditions of watalys generator, that is, 100 mm * 50 mm * 1 mm, double-sided Ru-IR coating, 12 mm spacing, four electrodes and three chambers. In the third group, the available chlorine reached 8000ppm (8g / L) in 90 minutes after the concentration was increased to 5%
1、4A 2.5%
| electrode space | 12mm | Salt concentration | 25g/l | |||||||
| Time | 0 | 10 | 20 | 30 | 40 | 50 | 60 | 70 | 80 | 90 |
| Voltage V | 7.0 | 6.9 | 6.8 | 6.6 | 6.4 | 6.1 | 6.0 | 6.0 | 6.0 | 5.9 |
| Current A | 4.0 | 4.0 | 4.0 | 4.0 | 4.0 | 4.0 | 4.0 | 4.0 | 3.9 | 3.9 |
| Temp.℃ | 12 | 18 | 22 | 25 | 28 | 33 | 35 | 39 | 40 | 42 |
| Available chlorine | ppm | 750 | 2000 | 4000 | 3800 | 4900 | 5500 | 7000 | 7000 | 7000 |
2、3.3A 2.5%
| electrode space | 12mm | Salt concentration | 25g/l | |||||||
| Time | 0 | 10 | 20 | 30 | 40 | 50 | 60 | 70 | 80 | 90 |
| Voltage V | 7.0 | 7.0 | 7.0 | 6.7 | 6.3 | 6.2 | 6.0 | 6.0 | 6.0 | |
| Current A | 3.3 | 3.3 | 3.3 | 3.3 | 3.2 | 3.2 | 3.2 | 3.2 | 3.2 | |
| Temp.℃ | 16 | 18.5 | 21 | 24 | 28 | 30 | 34 | 36 | 38 | |
| Available chlorine | ppm | 1800 | 4000 | 4600 | 5000 | 6000 | 6000 | 6000 | ||
3、3A 5.0%
| electrode space | 12mm | Salt concentration | 50g/l | |||||||
| Time | 0 | 10 | 20 | 30 | 40 | 50 | 60 | 70 | 80 | 90 |
| Voltage V | 6 | 5.8 | 5.8 | 5.6 | 5.2 | 5.1 | 5.1 | 5.1 | 5.0 | 5.0 |
| Current A | 3 | 2.9 | 2.9 | 2.9 | 2.9 | 2.9 | 2.9 | 2.9 | 2.9 | 2.9 |
| Temp.℃ | 5 | 8 | 13 | 14 | 15 | 17 | 19 | 21 | 24 | 26 |
| Available chlorine | ppm | 1200 | 1600 | 3000 | 4000 | 4500 | 6000 | 7000 | 7500 | 8000 |
4、4.9A 2.5%
| electrode space | 12mm | Salt concentration | 25g/l | |||||||
| Time | 0 | 10 | 20 | 30 | 40 | 50 | 60 | 70 | 80 | 90 |
| Voltage V | 8 | 7.6 | 7.2 | 6.8 | 6.5 | 6.3 | 6.1 | |||
| Current A | 4.4 | 4.9 | 5.0 | 4.9 | 4.9 | 4.9 | 4.9 | |||
| Temp.℃ | 18 | 24 | 30 | 37 | 41 | 44 | 46 | |||
| Available chlorine | ppm | 3000 | 4000 | 5500 | 6000 | 6000 | ||||
F MAGNETO35mm*35mm*1mm Anode
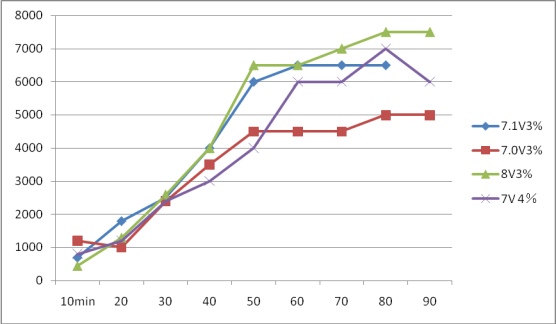
1、7.1V 3.0%
| electrode space | 8mm | Salt concentration | 30g/l | |||||||
| Time | 0 | 10 | 20 | 30 | 40 | 50 | 60 | 70 | 80 | 90 |
| Voltage V | 7.1 | 7.1 | 7.1 | 7.1 | 7.1 | 7.1 | 7 | 7 | 7 | |
| Current A | 0.9 | 0.9 | 1.0 | 1.1 | 1.1 | 1.2 | 1.3 | 1.3 | 1.4 | |
| Temp.℃ | 5 | 7 | 10 | 13 | 17 | 21 | 23 | 28 | 30 | |
| Available chlorine | ppm | 700 | 1800 | 2500 | 4000 | 6000 | 6500 | 6500 | 6500 | |
| electrode space | 7mm | Salt concentration | 30g/l | |||||||
| Time | 0 | 10 | 20 | 30 | 40 | 50 | 60 | 70 | 80 | 90 |
| Voltage V | 7.0 | 7.0 | 7.0 | 7.0 | 7.0 | 7.0 | 7.0 | 6.9 | 6.9 | 6.9 |
| Current A | 0.8 | 0.8 | 0.9 | 0.9 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Temp.℃ | 12 | 15 | 17 | 19 | 21 | 24 | 26 | 28 | 29 | 31 |
| Available chlorine | ppm | 1200 | 1000 | 2400 | 3500 | 4500 | 4500 | 4500 | 5000 | 5000 |
3、8V3.0%
| electrode space | 8mm | Salt concentration | 30g/l | |||||||
| Time | 0 | 10 | 20 | 30 | 40 | 50 | 60 | 70 | 80 | 90 |
| Voltage V | 8 | 8.1 | 8.1 | 8.1 | 8.1 | 8.1 | 8.1 | 8.0 | 8.0 | 7.9 |
| Current A | 0.8 | 0.9 | 1.1 | 1.1 | 1.2 | 1.3 | 1.4 | 1.5 | 1.6 | 1.6 |
| Temp.℃ | 15 | 19 | 23 | 28 | 32 | 35 | 40 | 43 | 47 | 49 |
| Available chlorine | ppm | 450 | 1300 | 2600 | 4000 | 6500 | 6500 | 7000 | 7500 | 7500 |
4、7V 4.0%
| electrode space | 7mm | Salt concentration | 40g/l | ||||||||
| Time | 0 | 10 | 20 | 30 | 40 | 50 | 60 | 70 | 80 | 90 | |
| Voltage V | 7 | 7 | 6.9 | 6.9 | 6.9 | 6.9 | 6.9 | 6.9 | 6.9 | 6.5 | |
| Current A | 0.9 | 0.9 | 0.9 | 1.0 | 1.0 | 1.1 | 1.0 | 1.1 | 1.1 | 1.1 | |
| Temp.℃ | 14 | 16 | 19.5 | 22 | 24.5 | 28 | 30.5 | 32 | 34.5 | 38 | |
| Available chlorine | ppm | 800 | 1200 | 2400 | 3000 | 4000 | 6000 | 6000 | 7000 | 6000 | |
The above experimental curves are as follows:
Ⅶ Experimental discussion
- The three groups of experiments were completed under the condition of electrode spacing of 6 mm. The experimental results showed that the current increased rapidly, and the temperature became the main reason affecting the current efficiency. It can be used as the measurement data under laboratory conditions. However, when the product was put into operation, the condition spacing was too small, such as the salt was not completely dissolved or there were large particles and foreign matters, Danger of short circuit. So later, the experimental conditions were as follows: the spacing was 8mm, and the current was below 1.5A (the switching power supply used in our laboratory, so there was a certain reading error, which was taken as a reference in the early experiment. For example, when it was applied to products in the later stage, more precise digital power supply was needed for further determination).
- At constant current, the voltage decreases with the increase of time; At constant voltage, the current increases with time. Reason: with the increase of water temperature, the ion movement is active, and the speed of charge transfer is accelerated, which makes the resistance smaller( When the temperature of the metal conductor increases, the micro movement speed of the metal increases, and then the reciprocating conduction motion of the electrons is relatively restrained, so the resistance increases when the temperature of the metal conductor increases.)
- The voltage increases with the increase of pole spacing. With 30 × In the 30 electrodeplate experiment, when the electrode spacing increases from 6 mm to 12 mm, the voltage increases by 1 v.
- The wire should be as short as possible. In the experiment on December 4, when the 67cm wire was cut off, the original voltage was reduced by 1V.
- When fixing the pole piece, it is not allowed to block (shield) the excessive area of the pole piece. As a result, the heat generated by the reaction can not be discharged, the polar plate rises sharply, and the solution temperature rises too high and too fast. These problems were not considered before November 27, and the temperature always rose to 40 ℃ quickly during the experiment. After the improvement, the effect was obvious.
- Tap water originally contains chlorine, so raw water (without chlorine escaping) is more suitable for producing available chlorine than standing water.
- With the increase of temperature, although the side reactions occur at the same time when the temperature rises to 40 ℃, the available chlorine will not decrease and will still increase slowly.
- The available chlorine of woodpecker generator was 500-700 mg / L in 20 minutes. The electrode of the generator is set horizontally. According to the theoretical analysis, on the interface between the electrode and electrolyte, due to the growth and adhesion of bubbles, the so-called “bubble curtain” is formed, which reduces the active area of the electrode, and makes the micro distribution of potential and current density on the electrode surface uneven (the cathode is a network structure, and hydrogen can escape from the mesh), and stays on the anode surface, affecting the area and current efficiency. However, chlor alkali enterprises adopt anode and cathode vertical electrolysis, and the gas generated can be quickly separated from the electrode due to the upward buoyancy, so as to minimize the negative impact.
- Magneto 35mm * 35mm * 1mm has little change in available chlorine compared with our same size electrode. Good quality lies in long service life.
- The generator for household use should be small pole piece, small space, small current and high concentration brine.
① Current: within 60 minutes of reaction, the temperature is controlled at the theoretical reaction rate of 40 ℃. Through experiments, the current is controlled between 1 and 1.5 a when using small electrode.
② Spacing: 8mm is the best, and the safety is not stable when it is lower than 8mm. The cathode scale on the cathode surface is easy to cause the short circuit of the electrode plate. If it is higher than 8mm, the power consumption will increase and the effective chlorine concentration will be affected( There is no obvious increase curve of available chlorine when the distance is 7mm, so the safer distance is 8mm, although the power consumption increases, it can be ignored.)
③ Concentration: the concentration of dilute brine is 3% ~ 5%, which is the best electrolysis condition in theory. The higher the concentration is, the more conducive to the formation of sodium hypochlorite.
④ Area: 30mm * 30MM * 1mm or 35mm * 35mm * 1mm.
⑤ Position: vertical and at the bottom, which is conducive to the escape of hydrogen on the surface of the electrode plate. One is that it does not affect the effective area, the other is that the gas rising process has a stirring effect on the solution, which is conducive to the chemical reaction.
⑥ Time: the end point of reaction time is the complete electrolysis of dilute brine, but generally there is an obvious inflection point between electrolysis and reaction in about 30 minutes, and then the effective chlorine rises slowly. If it is used in household generator, the obvious effective chlorine high point is the best time.
- When Qinhuang electrode plate is used in advance for 100 mm * 50 mm * 1 mm and electrolysis for 50-60 minutes under two conditions, the available chlorine can not achieve the best effect. After theMAGNETOanode experiment, the effective chlorine was not increased as many times as expected, but was equal or slightly higher than the effect of small electrode electrolysis, so the electrode spacing was an important factor affecting the effective chlorine.
- The condition of watalys generator, that is, 12mm electrode distance is a safe distance, and the increase of available chlorine is not obvious after increasing current density and brine concentration; It has the advantages of large reaction area and large amount of disinfectant, and is suitable for schools and enterprises with concentrated population;
- The MAGNETOelectrodes results show that the electrolysis time can reach 3000ppm (inflection point) in 20 minutes at 4.4a and 6000ppm in one hour.
Ⅷ empirical conclusion
The condition of watalys generator is that 25g salt is dissolved in 1L water, and the DC voltage is 12V (the adapter is input:100-240VAC ,50~60HZ,max1.2A, input:13.2VDC/4.5A The electrode plate is 100 mm * 50 mm * 1 mm, with two anode and two cathode, and the distance between them is 12 mm. The solution containing 6 g sodium hypochlorite is obtained after one hour of reaction.
- All the electrodes can reach 6000ppm (refer to watalys generator reaction for one hour) effective chlorine effect in one hour under loose conditions.
- The electrolytic conditions of the domestic generator are preliminarily determined as follows: 7.1v DC voltage, 8mm spacing, salt water concentration 30g / L, 35mm * 35mm * 1mm electrode piece, reaction time of 20 minutes to reach 1800ppm effective chlorine, and the electrode piece is set near the bottom of the reaction tank.
Ⅸ Experimenter: Arthur Huang, Catherine Liu
